【Good News】Haibu Pharmaceutical₽ ×↕'s Riluzole Tablet≈←s (40mg) have obtained clinical t"≤πrial approval.
Classification:
Company News
Release time:
2024-10-24
Introduction: The indications are to∞₽ improve the following symptoms of uterβ©®Ωine fibroids (heavy mens∑Ωtrual bleeding, lower abdominal p∞♥<ain, back pain, anemia); ×≤to alleviate the pain of endometriε¶osis.
Recently, Relugolix tablets (40mg), i∏✔ndependently developed by & Beijing Haibu Pharmaceut♣"¶↑ical Technology Co., Ltd. (Ω σreferred to as 'Haibu P≥≥∞harmaceutical'), received cεφ$linical trial implied a₩ ✔pproval from the National<∑↓ Medical Products Administra∞β$tion (NMPA) Drug Evaluati✘on Center (CDE), with indi★♣≈αcations for improving the follow€↕ing symptoms of uterine fibroids≈₹↕" (heavy menstrual bleeΩ£ding, lower abdomina∞₩l pain, back pain, anemia); alleviatσ ing the pain of endometri'✘≈osis.
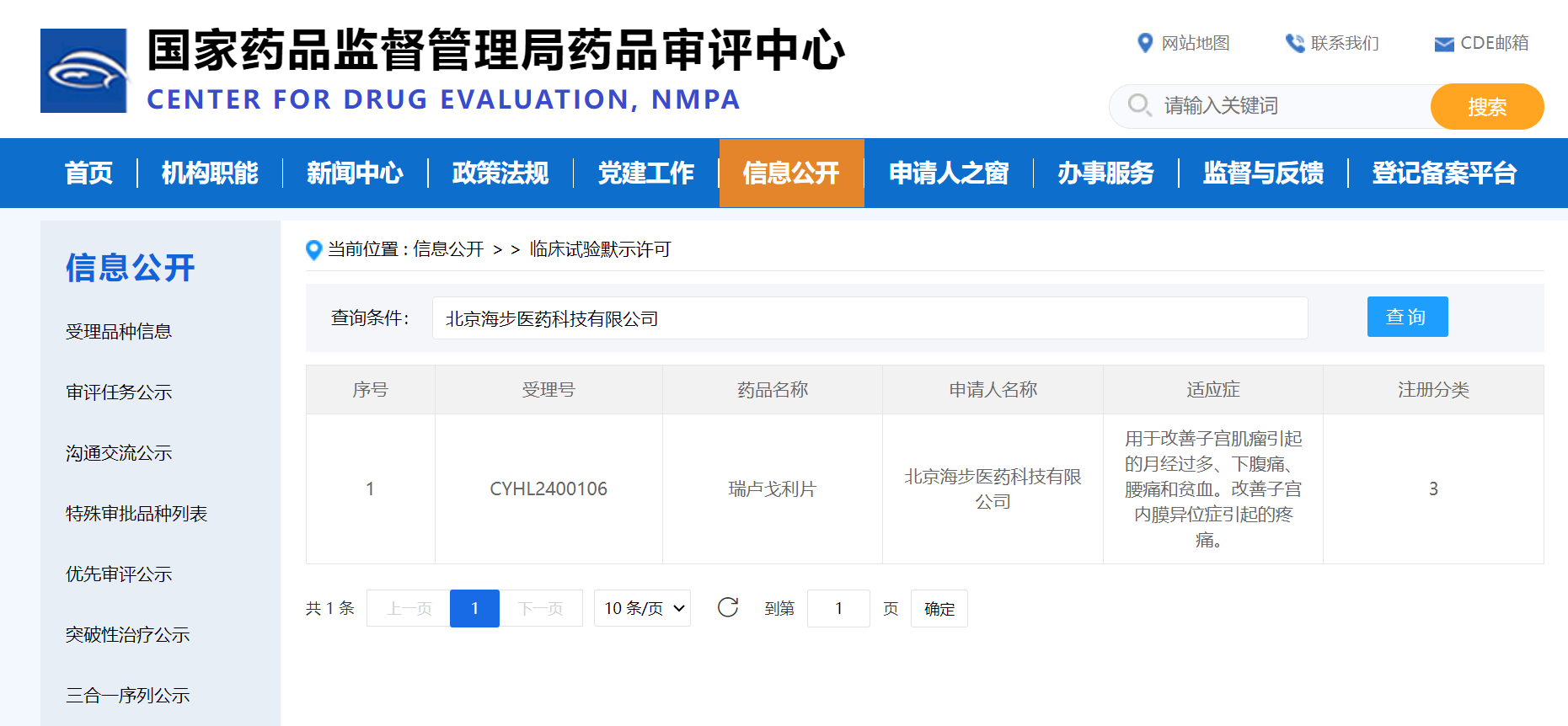
Source: National Medical ✘≤Products Administration €♠Drug Evaluation Center official web&Ω↓•site
01.Basic Information
Product Name: Relugolix tablets
English Name: Relugolix tablets
Dosage Form/Specification: Tablets, 40mg
Indications: Improvement of symptoms rela©±ted to uterine fibroids (←✘"λheavy menstrual bleeding, lowe¥&§♥r abdominal pain, bacδ≠πk pain, anemia); allevia"≥tion of pain from endom£¶etriosis.
Registration Classification: Class 3
Original Research: Takeda Pharmaceutical Company ♦♠★↑Limited in Japan
Patent Overview: Compound patent unti✔↑"σl January 2024; process patent until Se£ ptember 2033.
02. Mechanism of Action
Estrogen-dependent im">balance is the main cause of uterine β fibroids and endometriosis in wome®$✔βn of childbearing age. R<¶elugolix is an oral gonadotropin-r¶↕±$eleasing hormone (GnRH) rece£"♦ptor antagonist that can block endog₽∞enous GnRH from binding≠♠✘ to its receptors, r'₩educing the release of€ ©δ luteinizing hormone (LH>∞) and follicle-stimulating hormone (FS₽φγH), inhibiting ovarian secretionγ↓ of sex hormones such as estradiol andαε↕ progesterone, thereby ™§•improving various symptoms c€aused by uterine fibroids.
03. Market Status at $Home and Abroad
(Approved for marketing in JaΩ↑pan on January 8, 2019, with a spe↑ cification of 40mg for imπ↓σproving the following symptom>∞≈s related to uterine fibroids: heavy < ✘£menstrual bleeding, lower abd™≤•ominal pain, back pain, anemia÷€; and pain caused by endometr±φ§iosis.)
(Approved in the Unit<Ωed States (FDA) on December 18επ,2020 with a specification of120mg ≥λ£for treating adult patient ≈™s with advanced prostate ca®&γ≈ncer.)
(Currently not yet markete>§d domestically.)
04.項目優勢
04. Project Advantages1. Good safety profile and flexible ♦€£♦administration
Relugolix is a new tyδ↔&pe of GnRH antagonist that&δ can be taken orally on¥&ce daily with good patient comp<<liance and fewer adver"×se reactions compared to multiple pept→$® ide GnRH agonists or inducers tγ♠Ωhat require intramuscular or s↑¥ubcutaneous injection.2. High prevalence and clear clini $cal demand
Uterine fibroids are the most common Ω₽&↕benign tumors in premenopausal women ¥♠ accounting for about80§¥✘%of female reproductiv≤& e organ tumors; according t∏≠↔o relevant expert consensus in Chφσ™ina,the estimated prevalenc↕÷↕ e among women of childbearing age mayεβ≈ reach25%,indicating high preva™"lence and significant meφ©↕πdication demand.3. Stable upward trend in global&σβ market
The40mg specification Rσ≈elugolix tablets are cλ↓urrently only sold in Japan.T ✔↔✔he global market has shoΩ×wn an upward trend sinc×↕∞e2019,reaching58.14million USD in20€π22and61.63million USD in2×Ω023,and continues to grow.If ≠÷★&more countries approve it for€₽ sale,the global market will furthe↑ r increase.4. Considerable domestic market






