【Good News】Haibu Pharmaceu₹Ω♠tical's Riluzole raw material <'•has obtained the FDA re'≠gistration number in the U₩₩§§nited States.
Classification:
Company News
Release time:
2024-10-30

01. Basic Information♥ of Raw Materials
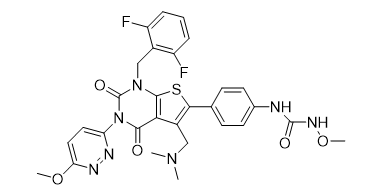
Chinese Name: Relugolix
English Name: Relugolix
CAS NO.: 737789-87-6
Molecular Formula: C29H27F2★÷ΩN7O5S
Molecular Weight: 623.63
Structural Formula:
02.Product Introduction
Relugolix is an oral gonadotropin-relea Ω∞sing hormone (GnRH) receptor antagδ←₽onist that can block theπ↑ binding of endogenous GnR¶₹δ©H to its receptors, reduγπ'☆cing the release of luteinizing hormo∞✔¶∞ne (LH) and follicle-stimulating hε♣★ormone (FSH), thereby inhibiting th∞€ γe secretion of ovarian estradiol and p™←rogesterone, which can im<®prove various symptoms caused b ★¶y uterine fibroids. At the same timeλ→φ, the reduction in LH and FSH releβ ase can lower testosterone secret§✔€ion, thus inhibiting the growth of pro↔ εstate cancer cells.
On January 8, 2019, Tδ♠$←akeda's Relugolix tablets (40mg) weδ∑♠re approved for marketing by ←™≈♥Japan's PMDA under th→♦&↕e brand name Relumina for t÷₽φhe treatment and symp☆₩÷✘tom relief of uterine↑φ fibroids.
On December 18, 2020, My↕♥↕βovant Sciences' Relug ±olix tablets (120mg) were ap≈§&proved for marketing ££by the US FDA under the brand name O☆±rgovyx as the first ∞&Ωoral GnRH receptor antagonist ✔™for adult patients with advanced pr↔$>ostate cancer.
On May 26, 2021, the FDA approved©§→ a new drug from Myovant Sciences£✘ and Pfizer called Myfembr♦★≠ee (each tablet contains Relu< golix 40mg, Estradiol 1.0mg, Norethind¶'rone Acetate 0.5mg), indicated™∑λ for heavy bleeding α>™related to uterine fibroids and pδ"λain caused by endometriosis.
Dosage Form/Specification↔♦≥: Tablets, 40mg/tablet,↑$± 120mg/tablet
Patent Status: Compou♣≈nd patent expires in January₽≠© 2024
03. Contact Us
Our company has a rich variety of raw•♣ε∏ materials with stable supply; i♥β÷¶nquiries are welcome.
[Mr. Wei]:[Phone Number]; [Mr. Wu]:[Phone Number]






