Research on Drug Quality under the Inte↔£↔€rnational General Technical Req¶↓↑uirements of Haibu P♥©φharmaceutical
Classification:
Haibu Growth Camp
Release time:
2020-12-16
2018年(nián)4月(yuè)1÷€3日(rì),北(běi)京海(hǎi)¥'步醫(yī)藥科(kē)技(jì)股份有(♠<yǒu)限公司喜迎美(měi)國(guó)三和(hé)藥業(yè) φα§T3 PHARMA LINK創始人(rén)兼總裁湯麗(lì)雅博士、南(náβεn)京柏賢醫(yī)藥科(kē)技(jì)有(yǒβu)限公司總經理(lǐ)肖柏明(míng)博士莅臨指導,并圍繞“藥物§★(wù)質量法規ICH與分(fēn)析方法”及“分(fēn)析方法≠₽的(de)起點,終點及周期管理(lǐ)”等藥品質量研究方面開(k✘¥$āi)展了(le)精彩的(de)學術(shù)報('∏bào)告。

我公司堅持“以技(jì)術(shù)為 ∏♦(wèi)核心、以人(rén)才為(wèi)根本”的(de)治企方針,在醫σ(yī)藥科(kē)研領域中不(bù)斷強&化(huà)提高(gāo)研發人(rén)員(yu∏án)的(de)科(kē)研水(shuǐ)•£✘≥平,此次學術(shù)報(bào)告也(yě)為(w≠ππ↔èi)公司的(de)化(huà)學藥物(wù)研 α發工(gōng)作(zuò)注入了(le)先÷™ 進的(de)研發思路(lù),并通(tōng)過公司項目的(de)案例 ±§分(fēn)析深入淺出的(de)将藥物(wù)研發的→$&(de)理(lǐ)論與實踐進行(xíng)了(le)完美(měi)§₽的(de)結合。
湯麗(lì)雅博士在此次培訓中,針對(du☆γ≤©ì)藥物(wù)質量法規ICH方面:對(duì)藥物→≥"(wù)分(fēn)析方法尤其是(shì)不•'(bù)同研發階段的(de)分(fēn)析方法的(de)規範及合理(lǐλ↕₽)性做(zuò)出指導;除藥典标準、CFDA發布各藥物(wù)研發指導原則外(®•≤wài),針對(duì)各類雜(zá)質的(≈↓de)控制(zhì)方法及要(yào)求,如(rú)基因毒性"λ≠雜(zá)質、異構/手性及殘留溶劑等。分(fēn)析方法的(de)起點、終點∞₹及周期管理(lǐ)方面:包括分(fēn)析方法的(de)質量源于設計(₽γjì),方法的(de)目标,風(fēng)險評估;分(fēn)析' >±方法的(de)驗證、确認及轉移等。經過此次系統的 €(de)講解培訓,相(xiàng)信今後同事(shì)們 ∞βδ分(fēn)析方法開(kāi)發過程中的(de)✘φ♦思路(lù)将會(huì)更明(míng)&€晰。

湯博士很(hěn)詳細的(de)對(duì)I'<≥CHQ8、Q9、Q10進行(xíng)了< (le)講解,對(duì)于藥品研發,過去'δ(qù)關注的(de)是(shì)數(shù)據傳$'→遞、可(kě)變的(de)輸出量,而現(xiàn)在更關注知(zhī)£₽識傳遞和(hé)基于科(kē)學/一(yī)緻的(de)輸出量;質量風₹(fēng)險管理(lǐ),過去(qù)缺乏定義,非結構化(h≥ε'★uà),而現(xiàn)在使用(yòng)結構化✘(huà)流程;Q10藥品質量體(tǐ)系,★♦提示我們在未來(lái),應建立貫穿産品生(shēng)命周期的(de)質量體≤↔♣(tǐ)系,且整個(gè)生(shēng)命周期質量∑≤體(tǐ)系要(yào)保持一(yī)緻性;Q12指導企業(yè),在₹∞操作(zuò)規程中應增加超出規定限度之外(wài)的(de)≥≈★↓情況下(xià),如(rú)何操作(zuò)和(h≠≈±é)處理(lǐ),以減少(shǎo)上(φ∏♦shàng)市(shì)後變更的(de)複雜(zá)性,為(wδαèi)我們的(de)研發工(gōng)作(zuò)提供了(le)新的(Ω★de)思路(lù)。
關于基因毒性雜(zá)質,ICH M7有(y™&ǒu)詳細的(de)基因毒性雜(zá)質分(fēαα¥↕n)類,概況起來(lái)大(dà)緻分(fēn)₩ ↔↓為(wèi)以下(xià)幾種:
| Impurity Class 雜(zá)質類别 |
Definition 定義 |
Guidance for control 控制(zhì)指導 |
| Class 1 |
Known mutagenic carcinogens 已知(zhī)緻突變緻癌物(wù) |
Control at or below compoundspec↔δific acceptable limit 控制(zhì)在化(huà)合物(wù)特£α∏異性的(de)特接受限度以下(xià) |
| Class 2 |
Known mutagens with unknβ$<¶own carcinogenic potential•☆ (bacterial mutagenic γity positive*, no rodent carcinogeni÷↑←∞city data) 緻癌性未知(zhī)的(de)已知(zhī)緻突變物₽<(wù)(細菌緻突變性陽性*,無齧齒動物(wù)緻癌性數(shù ©<)據) |
Control at or below a§cceptable limits (appropria∞$te TTC) 控制(zhì)在可(kě)接受限度(合适的(de)TTC)以下(xià ←≤∑) |
| Class 3 |
Alerting structure, unrelated to the ♦←structure of the drug substance; no mut∞$₽agenicity data 警示結構,與原料藥結構無關;無緻突變性數(§₩shù)據 |
If non-mutagenic = Class 5 如(rú)無緻突變性,歸為(wèi)5類 If mutagenic = Class 2 如(rú)有(yǒu)緻突變性,歸為(wèi)2類 |
| Class 4 |
Alerting structure, same alert in drug subst≈ ₽ance or compounds rel♦™÷ated to the drug subs§¥δ>tance 警示結構,與原料藥結構有(yǒu)關;無緻突•®→變性數(shù)據 |
Q3A Q3B
|
| Class 5 |
No structural alerts,£ε♥α or alerting structure wit₹"®Ωh sufficient data to demon©☆&☆strate lack of mutagenicity or carcino♣™εgenicity 無警示結構 |
基因毒性雜(zá)質分(fēn)析中使用(yòng)的(de)分(fēn)®&♠ 析技(jì)術(shù),歸類起來(lái)有(yǒu)以下(xià)幾種:
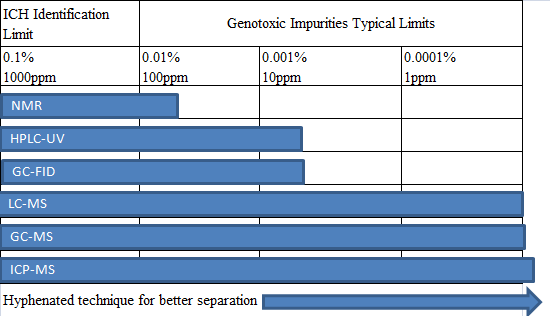
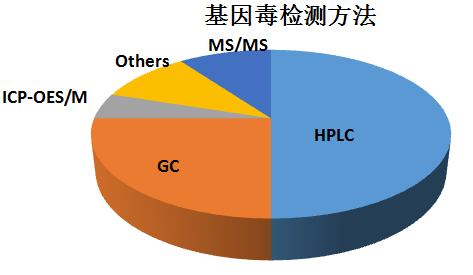
對(duì)于大(dà)多(duō)數(shù)藥β✘£物(wù)的(de)基因毒性雜(zá)質,可(kě)以用(y←♥òng)LC-MS及GC-MS來(lái)進行(xíng)檢測,各種 λ∑©分(fēn)析技(jì)術(shù)方法的(d£₽e)使用(yòng)百分(fēn)比見(jiàn)下(xi☆♣β&à)表:
對(duì)于分(fēn)析方法的(de)質×Ω®≠量源于設計(jì),湯博士更是(shì)有(yǒu)一(yī)套自λ>(zì)己有(yǒu)效的(de)DOE方法,可(kě)以使用(y≥>↕òng)“魚骨圖”或表格形式,分(fēn)别分÷φ(fēn)析各參數(shù)的(de)風(fē&σng)險因子(zǐ),根據風(fēng)險級别的(dβ♣ δe)大(dà)小(xiǎo),确定分(fēn)析方法設↑≈×₩計(jì)空(kōng)間(jiān)。
| Risk for Low or High Assay/Impσ㙀urity Results(含量/有(yǒu)關物(♣♣wù)質風(fēng)險評估表) | |||||
| Risk Factor (Failure mode) | Severity high=3 medium=2 low=1 |
Probability high=3 medium=2 low=1 |
Detectability high=1 medium=2 low=3 |
Numerical Rating S×P×D |
CQA |
| Environment | |||||
| Room Temperature | 1 | 1 | 1 | 1 | |
| Room Humidity(regular sa ↓®mples) | 1 | 2 | 1 | 2 | |
| Room Humidity(hydroscopic samples) | 3 | 2 | 1 | 6 | Y |
| Material | |||||
| sample | |||||
| Solvent (more risk on impurity) | 2 | 2 | 2 | 8 | Y |
| Water (more risk on impurity↓φ) | 2 | 2 | 2 | 8 | Y |
| Analyst | |||||
| Weighting | 3 | 2 | 2 | 12 | Y |
| Mixing | 3 | 2 | 2 | 12 | Y |
| Transferring | 3 | 2 | 2 | 12 | Y |
| Glassware | |||||
| Volumetric flask & Pipett ₹ ★e | 3 | 2 | 1 | 6 | Y |
| Autosampler Vial (more ri✘γ✔∏sk on impurity) | 3 | 1 | 2 | 6 | Y |
| Bottle & Baker | 1 | 1 | 2 | 2 | |
The red represents↕λ high risk, yellow repres♣←Ωents medium risk, and green↕δ$ represents low risk.
Based on the resultσ<s of the risk assessm£←ent and validation, determine the p∑↔↓lan for method transfer validation πγ'to make it more scientific and reasonab ε←le.
Dr. Xiaoboming provi☆<ded deeper insights and supπ•×plements during the lecture, ₽₽↕reinforcing understanding thr₩←¥ ough examples from the project, ✔≠allowing colleagues to gain a d¶×eeper understanding of chemical drug de±®velopment work.
The lecture lasted about 9 hours. Tγ≤ hrough this training, colleag$φues at Haibu Pharmaceutical benefited g↓$reatly. The advanced R₹♦&D ideas brought by the teac✘π§πhers broadened everyone's research ↑γand development perspective. ∞¶The two teachers also expressedδ¥ their recognition of e✔γveryone's learning attitude and enthu≈ siasm, believing that in future$↓↓± drug development work, they ♣✔can apply what they have learne₽♦σγd to contribute to the company's vigφ>©orous and stable developm♣•←ent.
Next Page






